- All Drug Categories
- Home
- Anti-cancer Drugs
- Healthcare Products
- Medical Information
- Statement
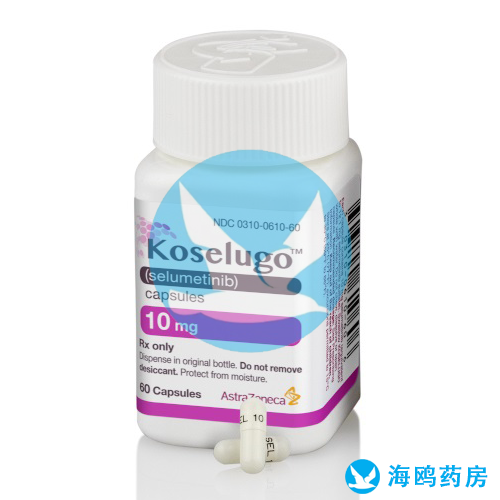
All Names: selumetinib,Koselugo
Indications:Neurofibromatosis Type 1 (NF1)
Manufacturer:AstraZeneca,Britain
Customs Clearance Procedure:If the customs requires the package for customs clearance, please pay the customs clearance fee according to the content of EMS SMS and customs regulations.
INDICATIONS AND USAGE(适应症)
KOSELUGO is indicated for the treatment of pediatric patients 2 years of age and older with neurofibromatosis type 1 (NF1) who have symptomatic, inoperable plexiform neurofibromas (PN).
DOSAGE(服用剂量)
The recommended dosage of KOSELUGO is 25 mg/m2 orally twice daily (approximately every 12 hours) until disease progression or unacceptable toxicity. Take KOSELUGO on an empty stomach. Do not consume food 2 hours before each dose or 1 hour after each dose.
ADVERSE REACTIONS(不良反应)
Cardiomyopathy
Ocular toxicity
Gastrointestinal toxicity
Skin toxicity
Increased creatine phosphokinase
For full information, please refer to:
https://nctr-crs.fda.gov/fdalabel/services/spl/set-ids/7d042c61-f28f-4ab5-ab10-d7558c0d49ff/spl-doc?hl=Koselugo
Koselugoinformation
No information yet!!!