- All Drug Categories
- Home
- Anti-cancer Drugs
- Healthcare Products
- Medical Information
- Statement
- Adults hospitalized for acute heart failure were 36% more likely to experience a clinical benefit over 90 days if initiated on Jardiance after ...

Shionogi Co. on Friday submitted an application for conditional early approval for Japan's first domestically developed COVID-19 pill, which...

OSAKA, Japan, and CAMBRIDGE, Massachusetts, February 28, 2022 – Takeda (TSE:4502/NYSE:TAK) today presented four abstracts including interim r...

The FDA released 3 new recommendations for industry to support and expand President Joe Biden’s 2016 Cancer Moonshot initiative. The new guid...
Phase 2 data highlighted a high complete response rate and undetectable minimal residual disease rate in patients with indolent clinical forms of...

The Food and Drug Administration (FDA) has approved Sage Chemical’s generic version of theApokyn(apomorphine hydrochloride injection) drug cartri...
The Food and Drug Administration (FDA) has revised the Emergency Use Authorization (EUA) for Evusheld (tixagevimab co-packaged with cilgavimab) t...
Targeted therapies, specifically those agents directed at mutated proteins and aberrant protein-to-protein interactions, have been shown ...
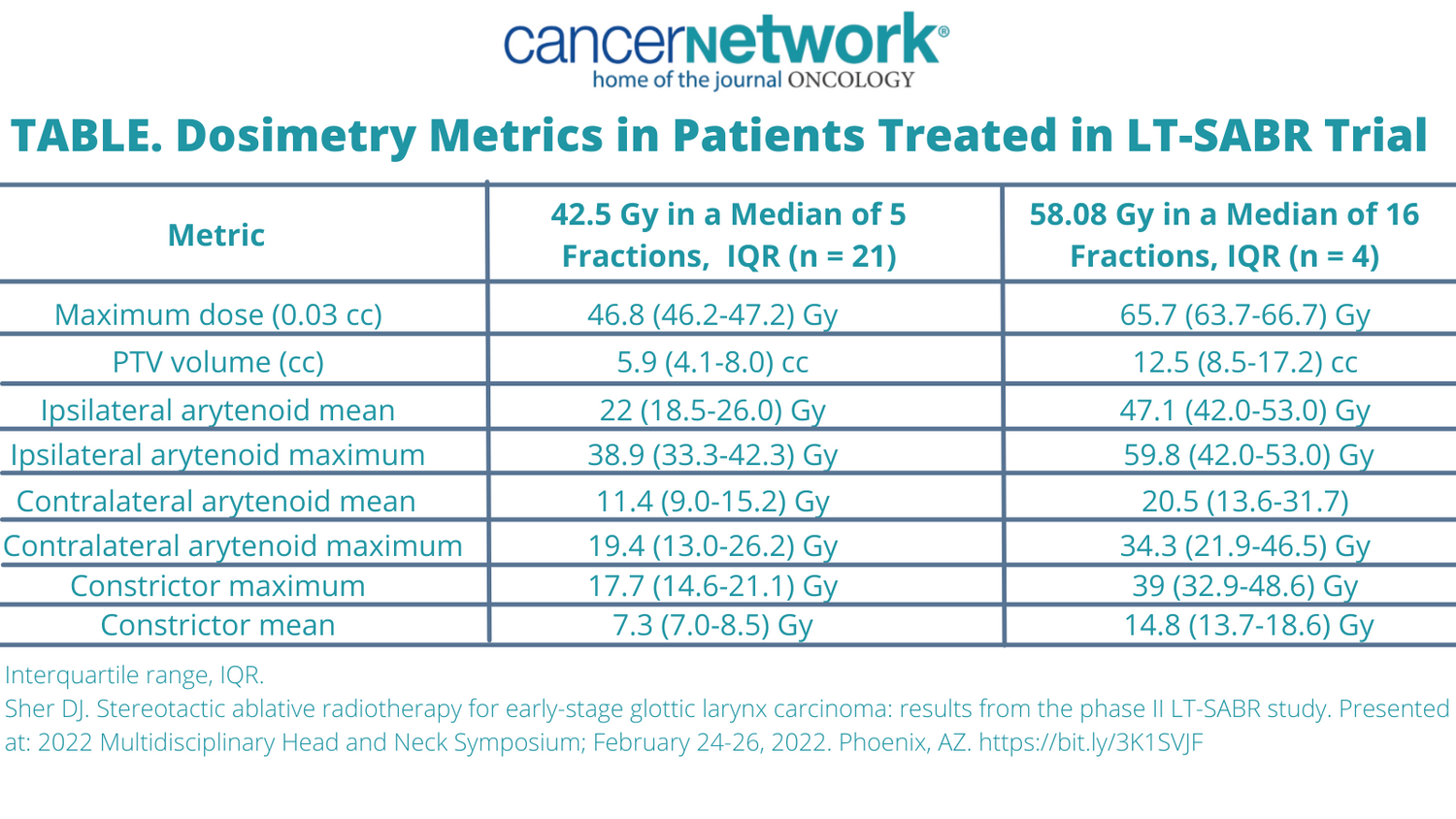
Treatment with tumor-focused stereotactic radiotherapy resulted in promising efficacy and patient reported outcomes (PROs) in those with early st...
BI 1015550 could treat both pulmonary fibrosis and inflammation associated with progressive fibrosing interstitial lung diseases. The FDA ha...