- All Drug Categories
- Home
- Anti-cancer Drugs
- Healthcare Products
- Medical Information
- Statement
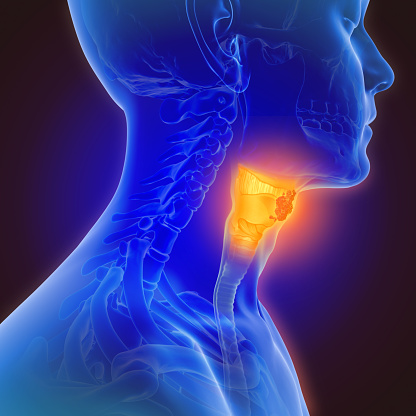
Treatment with tisotumab vedotin (Tivdak) resulted in promising safety and antitumor activity in a population of patients with squamous cell carc...
The Food and Drug Administration (FDA) has approved Releuko™ (filgrastim-ayow), a biosimilar to Neupogen® (filgrastim). The approval was based on...
HealthDay News — In a living systematic review and updated article published online March 1 in the Annals of Internal Medicine, final practice po...

The FDA has approved its first contact lens that comes equipped with its own antihistamine to help fight off eye itchiness caused by allergies du...

The addition of carboplatin to paclitaxel prolonged overall survival and progression-free survival, and proved to be noninferior to the standard ...

The MEK inhibitor trametinib ( Mekinist ) reduced the risk of disease progression or death by 52% compared with current standard-of-care (S...
NEW YORK--(BUSINESS WIRE)-- Pfizer Inc. (NYSE:PFE) today announced results from the CLOVER trial (CLOstridium difficileVaccineEfficacy TRial), a ...
Pacritinib (Vonjo) received accelerated approval from the FDA at a twice daily, 200-mg dose for patients with intermediate- or high-risk prim...
The European Medicines Agency's (EMA's) human medicines committee has recommended approval of a hepatitis B vaccine for adults. The agen...

The Committee for Medicinal Products for Human Use of the European Medicines Agency (EMA) has recommended approval of daridorexant (Quviviq, Idor...